Based on customer-reported outcomes
0x
Accelerated Project Setup
~0 min
Admin Time Recovered
~0 Emails
Reduced per Submission
0
Workspace for Your Team
Regulatory Impact Calculator
Quantify the operational impact of structured regulatory planning.
Traditional Spreadsheet Planning
Using SciFlow
26 min saved (72% faster)
Calculations are based on typical regulatory planning benchmarks and internal estimates. Actual impact may vary depending on organizational structure and submission complexity.
50+ purpose-built featuresfor regulatory teams
Purpose-built for regulatory teams. Every feature designed to eliminate manual work and accelerate submissions.
Version Control
Track every change with complete history. Roll back to any previous state with confidence.
Audit Trails
Complete audit trails designed to support 21 CFR Part 11 requirements and inspections.
In-Platform Collaboration
Project-specific communication keeps conversations organized and reduces email overload.
Regional Duplication
Clone entire projects across regions in seconds. Maintain consistency across global submissions.
Granular Permissions
Role-based access control with project-level security and enterprise authentication.
Live Dashboards
Real-time analytics provide instant visibility into capacity, timelines, and bottlenecks.
Project-specific dashboard
Visualize document status across the project (Planning, Draft, Under Review, Approved).
QC log (per project)
Track QC comments, resolutions, and completion status inside each project.
And more.
Built for Every Regulatory Role
Project Managers
- Centralized visibility across all projects
- Real-time status updates without manual tracking
- Better resource allocation & forecasting
- Reduced administrative burden
Regulatory Strategy
- Portfolio-level submission planning
- Strategic timeline visualization
- Multi-product scenario modeling
- Agency meeting planning
Regulatory Operations
- Structured eCTD content planning across the full lifecycle
- Built-in QC logging and audit-ready activity history
- Dynamic dashboards for sequence progress and operational metrics
- Live visibility into who is working on what — across portfolios
Organizations
- Eliminate Excel chaos & version conflicts
- No GxP validation required (Lower cost)
- Data-driven insights for planning
- Improved cross-functional collaboration
Visualize workyour way
Switch between Kanban, Gantt, Calendar, Portfolio, and Analytics views instantly. Every view syncs in real-time.
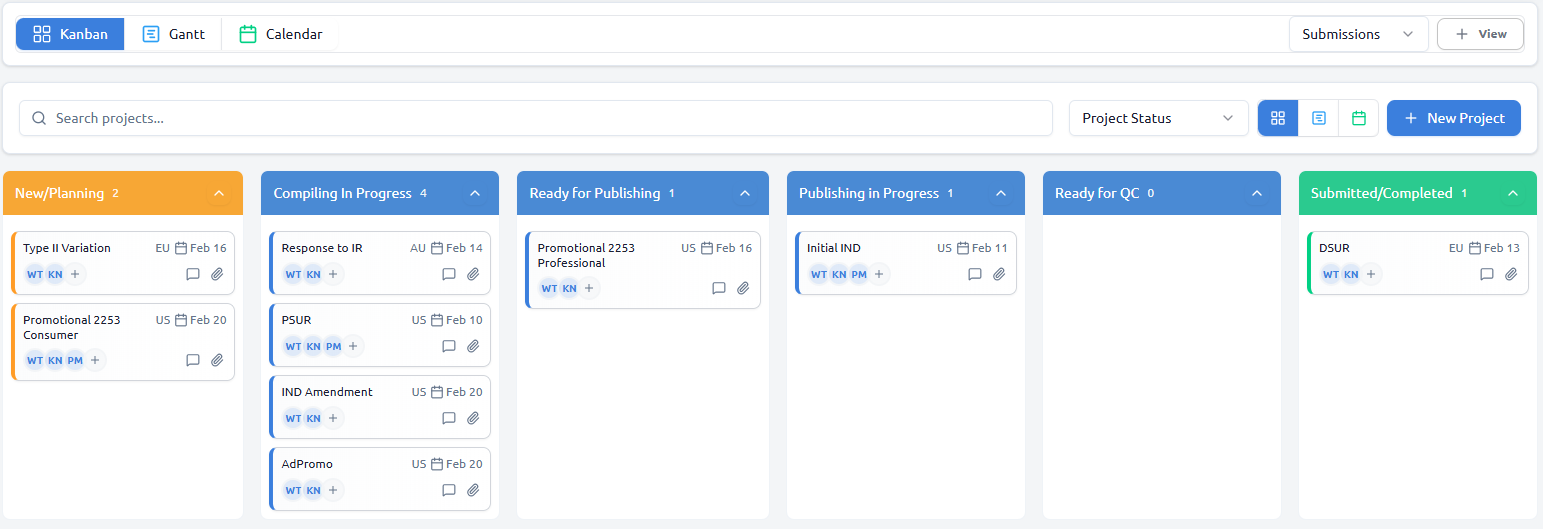
Kanban Board
Drag-and-drop cards across customizable status columns. Visualize bottlenecks and track submission progress at a glance.
- Customizable status workflows
- Real-time team synchronization
- Advanced filtering and search
Frequently Asked Questions
Answers to common questions about platform scope, governance, and implementation.